This is an old revision of the document!
Insight 46
Insight 46 is a neuroscience sub-study of the NSHD. Launched in 2015, Insight 46 recruited 502 study members selected at random from those who attended a clinical visit at 60–64 years and on whom relevant life course data are available. The study aims to investigate how life course factors and genetics influence the development of brain pathologies and impact on cognition, to provide insights into healthy brain ageing and the development of dementia.
For Insight 46, participants attended two clinic visits, two years apart. Each visit consists of a combination of brain imaging, biosampling and a clinical interview, including:
- Multi-modal MR imaging with concurrent acquisition of b-amyloid PET scanning.
- A neurological examination including assessment of gait and parkinsonian signs
- An enhanced cognitive assessment
- Sensitive tests of vision, hearing and smell
- Blood and urine sampling, and DNA collection
The second visit was extended to include an additional half day visit for more detailed cardiovascular testing and a lumbar puncture.
Combining these metrics with the extensive life course data already obtained from study members provides a unique opportunity to investigate the causes and consequences of various brain pathologies, to explore the differences between normal and abnormal brain ageing, and to identify a presymptomatic phase of dementia for future clinical trials aiming to prevent cognitive decline.
The Insight 46 recruitment and protocol papers are available to view.
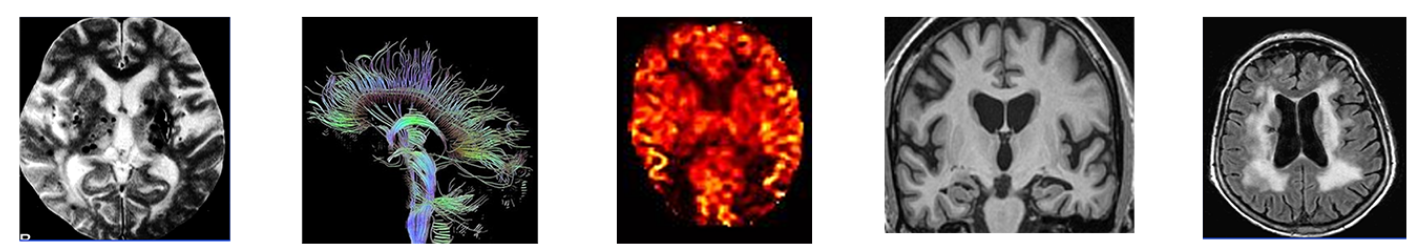
Accessing Insight 46 data
Most of the Insight 46 data collected at the first wave of clinic visits is available via Skylark, and access to these data can be requested in the same way as for the other NSHD variables.
The Insight 46 datasets currently include, but are not limited to:
- Amyloid status (positive/negative)
- Brain volumetric data, including volume of white matter hyperintensity
- Family history of neurological conditions
- Medication usage
Insight 46 was funded by Alzheimer’s Research UK, MRC Dementias Platform UK, The Wolfson Foundation, Weston Brain Institute, Brain Research UK, and British Heart Foundation. The 18F Florbetapir amyloid PET tracer was provided by AVID Radiopharmaceuticals, a wholly owned subsidiary of Eli Lilly.

